Quantitative Sciences Unit: It’s Not About the Sample Size
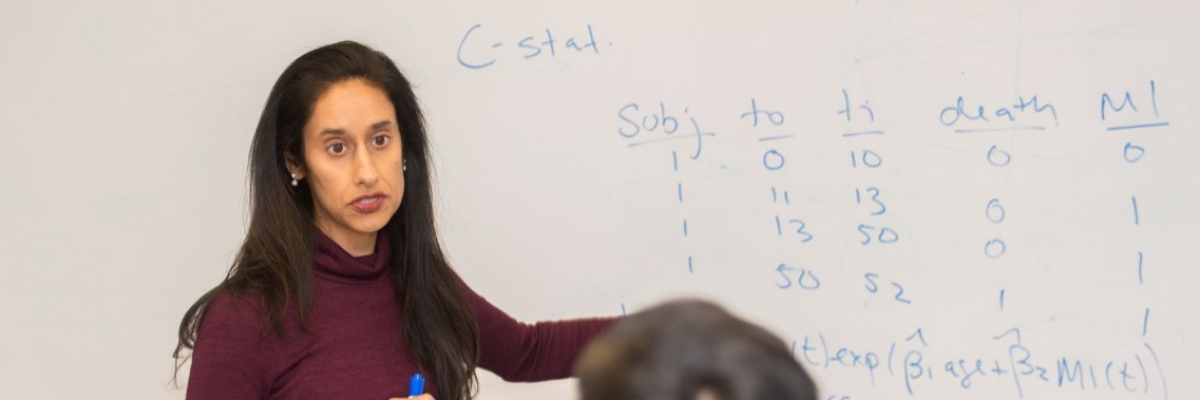
Manisha Desai, PHD, Professor of Biomedical Informatics Research
Quantitative Sciences Unit: It’s Not About the Sample Size
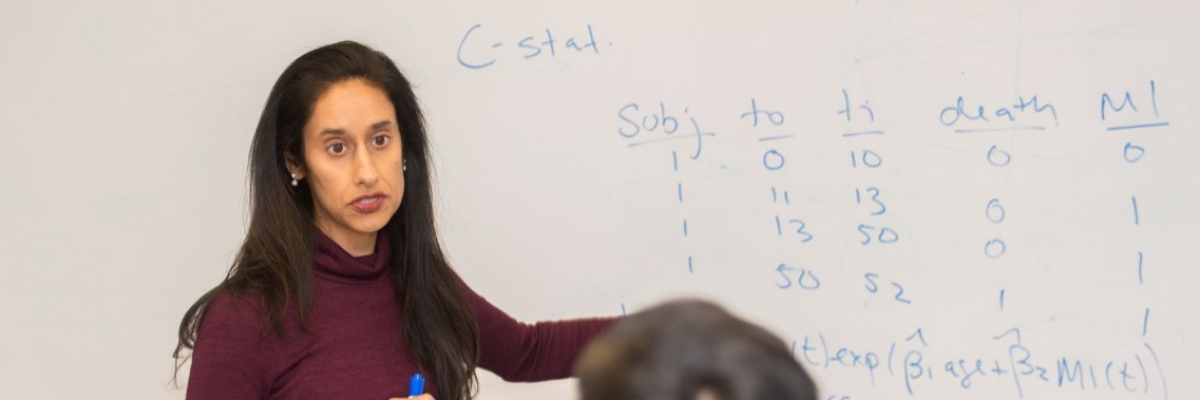
Manisha Desai, PHD, Professor of Biomedical Informatics Research
Quantitative Sciences Unit: It’s Not About the Sample Size
When Manisha Desai, PhD, a professor of biomedical informatics research, arrived at Stanford in 2009, she says she “kept hearing that there are just not enough statisticians on campus to provide all the necessary statistical support. And I felt that it shouldn’t be that way.”
There were some statistical groups, she noted, who were “wonderful at addressing consultative needs. When we started the Quantitative Sciences Unit (QSU), we wanted to make sure we complemented those statistical groups, which meant that we wanted to meet researchers’ needs with long-term collaborative partnerships. That’s really how we got established.”
First, there was a need to educate faculty in search of “just a sample size.” Desai talks about a typical scenario and how she changed it: “We got a lot of knocks on the door and someone would say, ‘I’ve got this grant; it’s due tomorrow. All I need is for you to bless it and give me the sample size calculation. I’m sure this will be quick and easy for you.’”
The education started immediately. Desai explains: “We had those people sit down and talk with us about their science: What are you trying to learn? What questions are you trying to address? We went back and forth about what’s known, what are the gaps, what are you trying to contribute scientifically. It’s a very different conversation than they were expecting to have.”
As that conversation continued, the dynamic changed. Desai goes on: “We showed them that we are actually scientists and can partner with them to help shape their questions, to make sure the questions are sensible and are getting at their goals. We also worked on refining hypotheses. Once all of that was done and we were on the same page, we talked about how best to design the set of experiments, the data to be generated that would be relevant for addressing the questions. Eventually, they began to see that this is a long iterative process. We would go back and forth, and that required scientific engagement. And now we write into NIH grant proposals that we need a biostatistical team for doing the data management and analyses and for partnering with the investigators.”
When Manisha Desai, PhD, a professor of biomedical informatics research, arrived at Stanford in 2009, she says she “kept hearing that there are just not enough statisticians on campus to provide all the necessary statistical support. And I felt that it shouldn’t be that way.”
There were some statistical groups, she noted, who were “wonderful at addressing consultative needs. When we started the Quantitative Sciences Unit (QSU), we wanted to make sure we complemented those statistical groups, which meant that we wanted to meet researchers’ needs with long-term collaborative partnerships. That’s really how we got established.”
First, there was a need to educate faculty in search of “just a sample size.” Desai talks about a typical scenario and how she changed it: “We got a lot of knocks on the door and someone would say, ‘I’ve got this grant; it’s due tomorrow. All I need is for you to bless it and give me the sample size calculation. I’m sure this will be quick and easy for you.’”
The education started immediately. Desai explains: “We had those people sit down and talk with us about their science: What are you trying to learn? What questions are you trying to address? We went back and forth about what’s known, what are the gaps, what are you trying to contribute scientifically. It’s a very different conversation than they were expecting to have.”
As that conversation continued, the dynamic changed. Desai goes on: “We showed them that we are actually scientists and can partner with them to help shape their questions, to make sure the questions are sensible and are getting at their goals. We also worked on refining hypotheses. Once all of that was done and we were on the same page, we talked about how best to design the set of experiments, the data to be generated that would be relevant for addressing the questions. Eventually, they began to see that this is a long iterative process. We would go back and forth, and that required scientific engagement. And now we write into NIH grant proposals that we need a biostatistical team for doing the data management and analyses and for partnering with the investigators.”
Sometimes investigators come to the QSU too late in the grant cycle for a proposal to be completed and successful. In those instances, Desai doesn’t hesitate to advise faculty to wait a cycle; in the current economic climate, such postponements have always proven to be advantageous for investigators. “They need to give it their best shot,” she says. “So in cases where people are really not ready, we encourage them to give us enough time to work together with them and show what we can bring to the table. We become a part of the team.”
In addition to spending a significant portion of their time collaborating on nascent and ongoing scientific projects, the QSU mentors faculty members who are new to research and are interested in learning the correct way to do their own studies. One such case is the Division of Hospital Medicine.
The QSU currently has 30 members, and five of them form an administrative core to triage new work. Desai explains that “we find out from our intake form how they came to our door and which department they are in. Depending on their resources and whether they need help with a grant proposal or unfunded data analyses, we figure out how to allocate our resources, how to prioritize the work, and then look for statistical expertise to match the need.”
While the teaching and collaborating take up a significant portion of the time available from the QSU, Desai stresses that “We are a research group, and we’re building our careers with those of our collaborators. And that’s the difference between consulting and collaborating. We are team members and coinvestigators, and we seek opportunities provided by our collaborators to lead research that is directly relevant and beneficial to them.”

We are a research group, and we’re BUILDING our careers with those our collaborators.
QSU Mentors Hospitalists in Research Methods
Since 2011, Neera Ahuja, MD, a clinical associate professor of hospital medicine, has grown her division from a faculty of seven to one of 36 in four distinct sections: surgical co-managers, hospitalists, nocturnists (who are hospitalists with overnight responsibility for inpatients), and Stanford Health Care–ValleyCare staff. With her faculty in place, she was ready to have them start doing research.
But, she realized, “We had only two or three faculty who had some research background, and we lacked biostatisticians. First we thought about hiring our own full-time biostatistician to have in our group. But Manisha [Desai, PhD, a biomedical informatics professor] very keenly said that person will feel isolated and won’t have the support of people who do what they do. So we partnered with the Quantitative Sciences Unit. Manisha was very open to a collaboration and in fact said that is what her group is meant to do because they are purely a research group. They want to support clinical groups like ours and find ways to guide and mentor. Now we fund a quarter of the salaries of two biostatisticians. Most of our research is quality improvement, medical education, and some informatics, where we have some biomedical informatics research experts run some data and do some analyses.”
Desai explains that the role of the QSU with young researchers such as the faculty in hospital medicine “has to do with mentoring them in research methods. We are partnering with Neera to help build up that research infrastructure. We want to help them understand such things as the grant submission process.”
Sometimes investigators come to the QSU too late in the grant cycle for a proposal to be completed and successful. In those instances, Desai doesn’t hesitate to advise faculty to wait a cycle; in the current economic climate, such postponements have always proven to be advantageous for investigators. “They need to give it their best shot,” she says. “So in cases where people are really not ready, we encourage them to give us enough time to work together with them and show what we can bring to the table. We become a part of the team.”
In addition to spending a significant portion of their time collaborating on nascent and ongoing scientific projects, the QSU mentors faculty members who are new to research and are interested in learning the correct way to do their own studies. One such case is the Division of Hospital Medicine.
The QSU currently has 30 members, and five of them form an administrative core to triage new work. Desai explains that “we find out from our intake form how they came to our door and which department they are in. Depending on their resources and whether they need help with a grant proposal or unfunded data analyses, we figure out how to allocate our resources, how to prioritize the work, and then look for statistical expertise to match the need.”
While the teaching and collaborating take up a significant portion of the time available from the QSU, Desai stresses that “We are a research group, and we’re building our careers with those of our collaborators. And that’s the difference between consulting and collaborating. We are team members and coinvestigators, and we seek opportunities provided by our collaborators to lead research that is directly relevant and beneficial to them.”

We are a research group, and we’re BUILDING our careers with those our collaborators.
QSU Mentors Hospitalists in Research Methods
Since 2011, Neera Ahuja, MD, a clinical associate professor of hospital medicine, has grown her division from a faculty of seven to one of 36 in four distinct sections: surgical co-managers, hospitalists, nocturnists (who are hospitalists with overnight responsibility for inpatients), and Stanford Health Care–ValleyCare staff. With her faculty in place, she was ready to have them start doing research.
But, she realized, “We had only two or three faculty who had some research background, and we lacked biostatisticians. First we thought about hiring our own full-time biostatistician to have in our group. But Manisha [Desai, PhD, a biomedical informatics professor] very keenly said that person will feel isolated and won’t have the support of people who do what they do. So we partnered with the Quantitative Sciences Unit. Manisha was very open to a collaboration and in fact said that is what her group is meant to do because they are purely a research group. They want to support clinical groups like ours and find ways to guide and mentor. Now we fund a quarter of the salaries of two biostatisticians. Most of our research is quality improvement, medical education, and some informatics, where we have some biomedical informatics research experts run some data and do some analyses.”
Desai explains that the role of the QSU with young researchers such as the faculty in hospital medicine “has to do with mentoring them in research methods. We are partnering with Neera to help build up that research infrastructure. We want to help them understand such things as the grant submission process.”




