Leading the Way in Esophageal Diseases
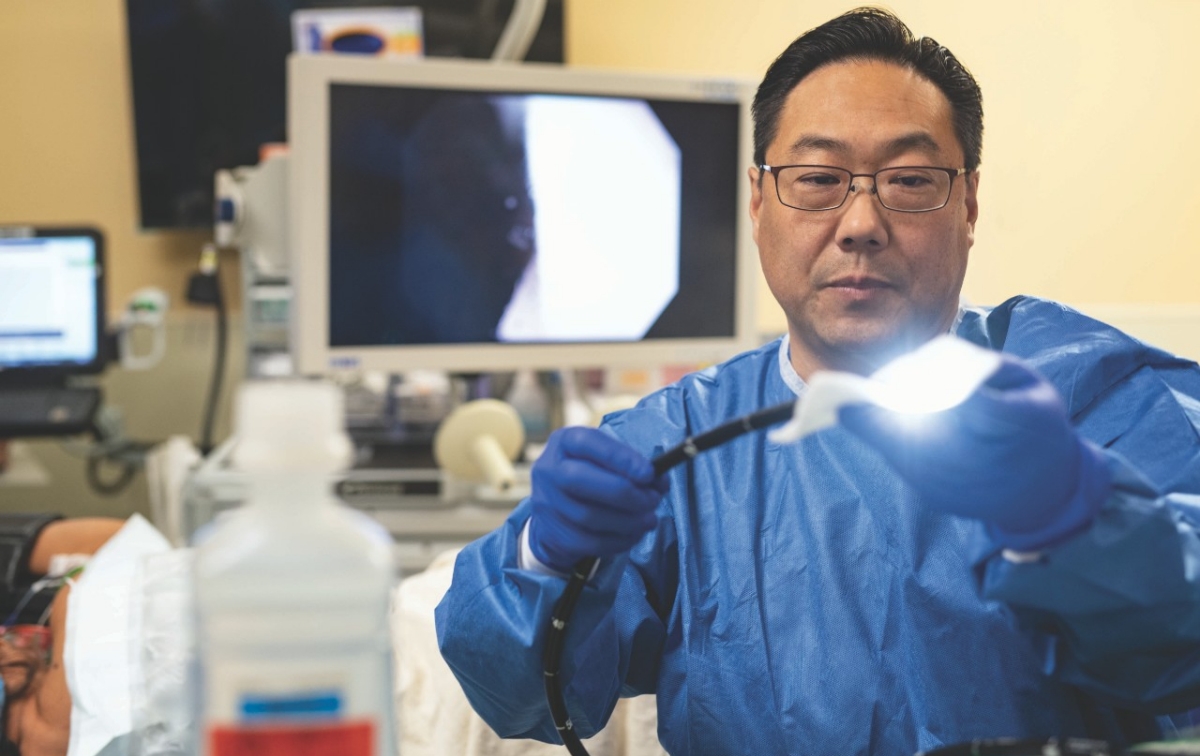
Joo Ha Hwang, MD, PhD, uses the endoscope to treat cancers and other gastrointestinal diseases.
Leading the Way in Esophageal Diseases

Joo Ha Hwang, MD, PhD, uses the endoscope to treat cancers and other gastrointestinal diseases.
Leading the Way in Esophageal Diseases
GASTROENTEROLOGIST JOO HA HWANG IS ONE OF JUST A FEW DOCTORS IN THE COUNTRY PERFORMING INNOVATIVE ENDOSCOPIC PROCEDURES TO TREAT CANCERS AND OTHER DISEASES OF THE ESOPHAGUS AND STOMACH.
Over the past decade, gastroenterologists in eastern Asia have perfected techniques that allow them to perform new procedures on the esophagus, stomach, and colon using an endoscope—a thin flexible tool that can snake through the digestive tract. The noninvasive techniques not only treat certain gastrointestinal diseases, but are helping lower the mortality rate from gastric cancer by offering a new, non-surgical way of removing precancerous and early cancerous lesions before they grow.
These experimental methods, however, have been slow to catch on in the United States. At Stanford, gastroenterologist Joo Ha Hwang, MD, PhD, professor of medicine, wants to change that.
Hwang is leading the way in performing and studying these new esophageal approaches and advocating for better gastric cancer screening. “These procedures, which we’re helping to further develop and study, really represent the cutting-edge medicine that we’re delivering here at Stanford,” says Hwang.
An At-Risk Population
Gastric cancer is the fifth most common cancer worldwide, but its incidence in the United States is relatively low—about 27,000 people are diagnosed with stomach cancer each year. Its prevalence is highest in people of Asian, Pacific Islander, and Hispanic descent, but even among these populations, there are no official screening guidelines in this country.
“It tends to be diagnosed at a late stage here, because the early stage cancer often doesn’t have symptoms,” says Hwang. “That means the survival rate is not very good.”
n the United States, physicians recommend that adults get screening colonoscopies, in which an endoscope is used to look at the lower part of the gastrointestinal tract, where colon cancer can occur. But this lower endoscopy doesn’t cover the stomach and other “upper” areas of the digestive system.
In Asia, however, gastroenterologists use an endoscope to examine the upper part of the gastrointestinal tract for signs of early cancer in adults every few years. That means most cases of gastric cancer there are identified much earlier, and the survival rate in Asia is better than in the United States.
Hwang and his colleagues have been studying the incidence of gastric cancer in the United States and the most at-risk populations, and think that regular screening within certain populations could help reduce mortality here.
GASTROENTEROLOGIST JOO HA HWANG IS ONE OF JUST A FEW DOCTORS IN THE COUNTRY PERFORMING INNOVATIVE ENDOSCOPIC PROCEDURES TO TREAT CANCERS AND OTHER DISEASES OF THE ESOPHAGUS AND STOMACH.
Over the past decade, gastroenterologists in eastern Asia have perfected techniques that allow them to perform new procedures on the esophagus, stomach, and colon using an endoscope—a thin flexible tool that can snake through the digestive tract. The noninvasive techniques not only treat certain gastrointestinal diseases, but are helping lower the mortality rate from gastric cancer by offering a new, non-surgical way of removing precancerous and early cancerous lesions before they grow.
These experimental methods, however, have been slow to catch on in the United States. At Stanford, gastroenterologist Joo Ha Hwang, MD, PhD, professor of medicine, wants to change that. Hwang is leading the way in performing and studying these new esophageal approaches and advocating for better gastric cancer screening. “These procedures, which we’re helping to further develop and study, really represent the cutting-edge medicine that we’re delivering here at Stanford,” says Hwang.
An At-Risk Population
Gastric cancer is the fifth most common cancer worldwide, but its incidence in the United States is relatively low—about 27,000 people are diagnosed with stomach cancer each year. Its prevalence is highest in people of Asian, Pacific Islander, and Hispanic descent, but even among these populations, there are no official screening guidelines in this country.
“It tends to be diagnosed at a late stage here, because the early stage cancer often doesn’t have symptoms,” says Hwang. “That means the survival rate is not very good.”
In the United States, physicians recommend that adults get screening colonoscopies, in which an endoscope is used to look at the lower part of the gastrointestinal tract, where colon cancer can occur. But this lower endoscopy doesn’t cover the stomach and other “upper” areas of the digestive system.
In Asia, however, gastroenterologists use an endoscope to examine the upper part of the gastrointestinal tract for signs of early cancer in adults every few years. That means most cases of gastric cancer there are identified much earlier, and the survival rate in Asia is better than in the United States.
Hwang and his colleagues have been studying the incidence of gastric cancer in the United States and the most at-risk populations, and think that regular screening within certain populations could help reduce mortality here. In 2020, they’ll be hosting a gastric cancer summit at Stanford. “We’re bringing in leaders in gastric cancer from all over the world to talk about how we can establish screening guidelines for high-risk populations in the U.S.,” says Hwang.
Less Invasive Surgery
The push toward increased gastric cancer screening is, in part, due to a relatively new procedure to help remove pre-cancerous and early cancerous lesions from the stomach. Until recently, such lesions could be seen and biopsied using the endoscope, but removing them required surgery.
Now, specially trained gastroenterologists—like Hwang, who went to Korea and Japan for training—can surgically remove the pre-cancers and early cancers endoscopically. The procedure, called an endoscopic submucosal dissection (ESD), takes only a few hours. Patients can generally go home the following day, rather than staying in the hospital several nights as they might after a typical surgery.
“Stanford is now one of the highest volume sites in the United States for ESD,” says Hwang. “We get patients not only from Northern California, but from Hawaii, Alaska, and really all over the country.”
There’s another procedure in the upper digestive tract that’s also heralding a shift from more invasive surgeries toward endoscopic versions. It’s called per-oral endoscopic myotomy, or POEM, and it involves cutting into the muscles that surround the esophagus. Like with ESD, Hwang learned the approach abroad and is now one of the leading POEM providers in the United States. He uses it to treat two conditions—achalasia and gastroparesis.

Stanford is now one of the highest volume sites in the United States for ESD
In achalasia, patients have trouble swallowing because the ring-shaped muscle between the esophagus and stomach doesn’t relax normally, trapping food in the esophagus. Before POEM, the mainstay treatments for achalasia were Botox injections—which relax the muscles, but only for a few months—or surgery.
In POEM, Hwang can use a small knife passed through an endoscope to cut through the inner layers of the esophagus to access the muscle and make a slit in it. Then the esophagus is closed with tiny clips.
“Patients can’t feel the incision or clips, and they can swallow right away. It’s one of the most gratifying procedures I do,” says Hwang.
In another version of POEM, known as gastric per-oral endoscopic myotomy, or G-POEM, the same approach is used to cut the muscle at the base of the stomach, the pylorus, for people with gastroparesis. In this more common disease, the stomach can’t empty itself of food in a normal fashion, leading to heartburn, nausea, vomiting, and feeling full quickly when eating.
Collaborative Research
As the number of esophageal endoscopy procedures offered at Stanford grows, the Esophagus Center is becoming a dynamic epicenter for collaboration and research. Hwang works with other gastroenterologists as well as surgeons and otolaryngologists.
“We are very integrated with the surgery department, and it’s a very multidisciplinary collaborative group,” says Hwang. He’s also working closely with the Stanford Medicine Center for Asian Health Research and Education (CARE) on his gastric cancer screening efforts.
Research questions remain, such as which patient populations benefit most from ESD and POEM, and what are the underlying causes of achalasia and gastroparesis. During POEM procedures, Hwang takes biopsies that may help answer these questions.
The numbers of ESD and POEM cases at Stanford are some of the highest in the country. There are very few training opportunities in the United States for physicians to learn these cutting-edge procedures. Hwang hopes that changes in the near future: “We hope to offer a fellowship training program in these eventually, to help disseminate the knowledge and techniques so that other patients may benefit from these procedures.”

Stanford is now one of the highest volume sites in the United States for ESD
In 2020, they’ll be hosting a gastric cancer summit at Stanford. “We’re bringing in leaders in gastric cancer from all over the world to talk about how we can establish screening guidelines for high-risk populations in the U.S.,” says Hwang.
Less Invasive Surgery
The push toward increased gastric cancer screening is, in part, due to a relatively new procedure to help remove pre-cancerous and early cancerous lesions from the stomach. Until recently, such lesions could be seen and biopsied using the endoscope, but removing them required surgery.
Now, specially trained gastroenterologists—like Hwang, who went to Korea and Japan for training—can surgically remove the pre-cancers and early cancers endoscopically. The procedure, called an endoscopic submucosal dissection (ESD), takes only a few hours. Patients can generally go home the following day, rather than staying in the hospital several nights as they might after a typical surgery.
“Stanford is now one of the highest volume sites in the United States for ESD,” says Hwang. “We get patients not only from Northern California, but from Hawaii, Alaska, and really all over the country.”
There’s another procedure in the upper digestive tract that’s also heralding a shift from more invasive surgeries toward endoscopic versions. It’s called per-oral endoscopic myotomy, or POEM, and it involves cutting into the muscles that surround the esophagus. Like with ESD, Hwang learned the approach abroad and is now one of the leading POEM providers in the United States. He uses it to treat two conditions—achalasia and gastroparesis.
In achalasia, patients have trouble swallowing because the ring-shaped muscle between the esophagus and stomach doesn’t relax normally, trapping food in the esophagus. Before POEM, the mainstay treatments for achalasia were Botox injections—which relax the muscles, but only for a few months—or surgery.
In POEM, Hwang can use a small knife passed through an endoscope to cut through the inner layers of the esophagus to access the muscle and make a slit in it. Then the esophagus is closed with tiny clips.
“Patients can’t feel the incision or clips, and they can swallow right away. It’s one of the most gratifying procedures I do,” says Hwang.
In another version of POEM, known as gastric per-oral endoscopic myotomy, or G-POEM, the same approach is used to cut the muscle at the base of the stomach, the pylorus, for people with gastroparesis. In this more common disease, the stomach can’t empty itself of food in a normal fashion, leading to heartburn, nausea, vomiting, and feeling full quickly when eating.
Collaborative Research
As the number of esophageal endoscopy procedures offered at Stanford grows, the Esophagus Center is becoming a dynamic epicenter for collaboration and research. Hwang works with other gastroenterologists as well as surgeons and otolaryngologists.
“We are very integrated with the surgery department, and it’s a very multidisciplinary collaborative group,” says Hwang. He’s also working closely with the Stanford Medicine Center for Asian Health Research and Education (CARE) on his gastric cancer screening efforts.
Research questions remain, such as which patient populations benefit most from ESD and POEM, and what are the underlying causes of achalasia and gastroparesis. During POEM procedures, Hwang takes biopsies that may help answer these questions.
The numbers of ESD and POEM cases at Stanford are some of the highest in the country. There are very few training opportunities in the United States for physicians to learn these cutting-edge procedures. Hwang hopes that changes in the near future: “We hope to offer a fellowship training program in these eventually, to help disseminate the knowledge and techniques so that other patients may benefit from these procedures.”