From Bariatric Surgeon to Accidental Activist

Arghavan Salles, MD, PhD
From Bariatric Surgeon to Accidental Activist
How One MD/PhD Harnesses the Power of Social Media for Advocacy and Career Development

Arghavan Salles, MD, PhD
From Bariatric Surgeon to Accidental Activist
How One MD/PhD Harnesses the Power of Social Media for Advocacy and Career Development
Arghavan Salles, MD, PhD, did not set out to be an activist or social media influencer. After completing medical school, a surgery residency, and a PhD in education at Stanford, Salles worked as a bariatric surgeon and faculty member in St. Louis, Missouri. She now looks back on the toll those years of grueling medical service and an unsuccessful fertility journey took on her energy. In 2019, she returned to Stanford and the Bay Area for a change of scene and an opportunity to renew her energy and drive.
Now, Salles is a clinical associate professor of gastroenterology and hepatology and special adviser of diversity, equity, and inclusion programs in the Department of Medicine. She found renewed motivation in an unexpected place: Twitter, which she joined in 2016.
She initially shared academic posts (research papers and bariatric surgery content, for example). Over time, she shared more personal content grounded in her sense of justice and responsibility.
Salles identifies, in her own words, as “an academic physician who does research to shed light on certain problems and push our institutions and society toward more inclusive policies” and uses social media primarily as a useful tool in that context.
Below, Salles reflects on the energy driving her posts and how to use social media as a tool for both advocacy and career advancement.

People say silence is complicity, and I do think that is true to some extent. The more we ignore things, the more we suggest those things are fine.
What drives you to do the work of social media activism each day?
I think what keeps me going is a little bit of naïveté — feeling like we can make a difference, that more people speaking up can shift culture and open people’s eyes to issues.
There’s also a feeling of discomfort and uncertainty. I feel very unsettled about a lot of the things happening in the world. I can’t just sit on the sidelines and be like, Oh, someone else will figure that out. Or, It’s OK if I don’t say anything. The more we ignore things, the more we suggest those things are fine.
I have a very strong sense of justice, which may not align with someone else’s, but it is all mine. What I think is right is something I often want to fight for and speak up about. That desire doesn’t really fade from day to day.
Is there a post that stands out as especially representative of your work?
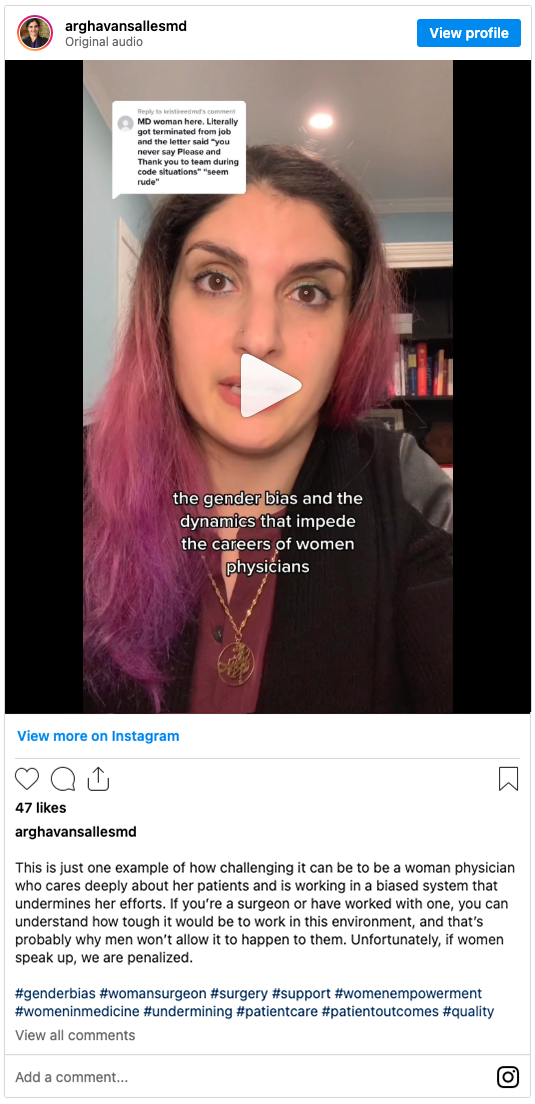
I had this video in January that was about gender schemas, particularly how we view women’s behavior differently just because they’re women, especially in male-dominated professions and anytime we have to be in authoritative roles. There were many women who were like, This is happening every day of my life. They didn’t know the research around this, so my post was validating for them.
There’s a lot we don’t talk about publicly. That leaves people feeling alone and isolated. Even though what they are experiencing is something that’s relatively common, it’s not talked about. I try to communicate to the people who need to hear it: If you’re experiencing this, know that other people experience it too. That doesn’t mean it’s OK, and we should try to fix the systems in place that make this a reality. But also, you’re not alone.
A lot of what I’ve been doing in the last year or so has been about pushing the boundaries of what we think of as professionalism. In most of our organizations, professionalism is weaponized against marginalized people. And it’s really all just made up.
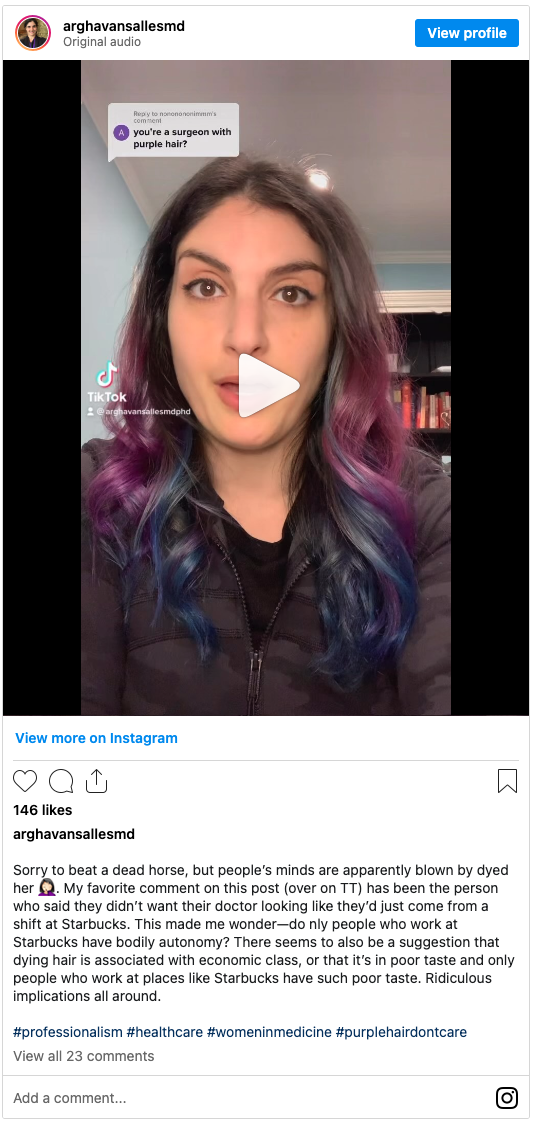
For example, in an Instagram reel I posted in March about being a surgeon with hyper-colored hair, I talk about how I can show up to a place, wearing clean clothes, having showered, having my hair done, whatever, and just because the color of my hair is different than what people are used to, that makes me not professional. So I believe that people should have autonomy over their bodies. It’s not harming anyone else, and it’s not affecting their ability to do their jobs.
What advice would you offer to academics interested in becoming more active on social media, and perhaps in doing advocacy on social platforms?
It’s such an interesting time. Two years ago, I might have said everyone should be on Twitter. But Twitter [rebranded as X in July 2023] is evolving, right? Not necessarily in a positive direction, so it’s hard for me to say that now.
But what I can say is that those of us who are on Twitter have developed collaborations from being in that space. We’ve made friends, and we’ve built community. I’ve found mentors through Twitter who I wouldn’t have met otherwise. I met most of the collaborators on my R01 grant, “Sexual Harassment Training of Primary Investigators (STOP),” via social media.
Social media can be a powerful tool for career development, especially when people are thinking about promotions to associate or full professor, and it can help in developing a regional or national reputation (especially for people who don’t focus on academic publishing).
I don’t think it has to be about activism for everyone. There’s value in networking and in accessing information and scientific research. I think social media is more effective for keeping up on the latest science than going from journal to journal or newspaper to newspaper. Information on research, clinical trials, and the latest things you need to know about the practice of medicine is much more accessible on social media.


People say silence is complicity, and I do think that is true to some extent. The more we ignore things, the more we suggest those things are fine.
Salles identifies, in her own words, as “an academic physician who does research to shed light on certain problems and push our institutions and society toward more inclusive policies” and uses social media primarily as “a useful tool in that context.”
Below, Salles reflects on the energy driving her posts and how to use social media as a tool for both advocacy and career advancement.
What drives you to do the work of social media activism each day?
I think what keeps me going is a little bit of naïveté — feeling like we can make a difference, that more people speaking up can shift culture and open people’s eyes to issues.
There’s also a feeling of discomfort and uncertainty. I feel very unsettled about a lot of the things happening in the world. I can’t just sit on the sidelines and be like, Oh, someone else will figure that out. Or, It’s OK if I don’t say anything. The more we ignore things, the more we suggest those things are fine.
I have a very strong sense of justice, which may not align with someone else’s, but it is all mine. What I think is right is something I often want to fight for and speak up about. That desire doesn’t really fade from day to day.
Is there a post that stands out as especially representative of your work?
I had this video in January that was about gender schemas, particularly how we view women’s behavior differently just because they’re women, especially in male-dominated professions and anytime we have to be in authoritative roles. There were many women who were like, This is happening every day of my life. They didn’t know the research around this, so my post was validating for them.
There’s a lot we don’t talk about publicly. That leaves people feeling alone and isolated. Even though what they are experiencing is something that’s relatively common, it’s not talked about. I try to communicate to the people who need to hear it: If you’re experiencing this, know that other people experience it too. That doesn’t mean it’s OK, and we should try to fix the systems in place that make this a reality. But also, you’re not alone.
A lot of what I’ve been doing in the last year or so has been about pushing the boundaries of what we think of as professionalism. In most of our organizations, professionalism is weaponized against marginalized people. And it’s really all just made up.
For example, in an Instagram reel I posted in March about being a surgeon with hyper-colored hair, I talk about how I can show up to a place, wearing clean clothes, having showered, having my hair done, whatever, and just because the color of my hair is different than what people are used to, that makes me not professional. So I believe that people should have autonomy over their bodies. It’s not harming anyone else, and it’s not affecting their ability to do their jobs.
What advice would you offer to academics interested in becoming more active on social media, and perhaps in doing advocacy on social platforms?
It’s such an interesting time. Two years ago, I might have said everyone should be on Twitter. But Twitter [rebranded as X in July 2023] is evolving, right? Not necessarily in a positive direction, so it’s hard for me to say that now.
But what I can say is that those of us who are on Twitter have developed collaborations from being in that space. We’ve made friends, and we’ve built community. I’ve found mentors through Twitter who I wouldn’t have met otherwise. I met most of the collaborators on my R01 grant, “Sexual Harassment Training of Primary Investigators (STOP),” via social media.
Social media can be a powerful tool for career development, especially when people are thinking about promotions to associate or full professor, and it can help in developing a regional or national reputation (especially for people who don’t focus on academic publishing).
I don’t think it has to be about activism for everyone. There’s value in networking and in accessing information and scientific research. I think social media is more effective for keeping up on the latest science than going from journal to journal or newspaper to newspaper. Information on research, clinical trials, and the latest things you need to know about the practice of medicine is much more accessible on social media.